Fully solved answers to the NCERT textbook exercise of Chapter 11, from short reasoning parts to numericals on heat, work, internal energy, entropy and heat engines, written in clear, exam‑oriented steps.[web:27][web:30]
Initial questions focus on thermodynamic systems, equilibrium, state variables and processes (isothermal, adiabatic, isochoric, cyclic), explaining each answer using diagrams and precise textbook language so you can mirror them in exams.[web:27][web:31]
The second part applies the first law of thermodynamics, work from \(P\!-\!V\) graphs, heat engines and efficiency to compute heat exchange, internal‑energy change and work done, with units and sign conventions highlighted at every step.[web:27][web:36]
Thermodynamics usually contributes multiple questions across JEE papers, and NCERT‑style exercise problems on work, heat and cyclic processes form the base for many objective and integer‑type questions.[web:36]
NEET focuses on straightforward calculations and conceptual clarity in topics like heat engines and specific heat, so mastering each NCERT exercise part gives direct scoring opportunities.[web:31][web:36]
Board papers and school tests often adapt directly from the NCERT exercise, and olympiad‑style questions frequently extend these same ideas on entropy, reversibility and engine efficiency.[web:27][web:33]
Q1. A geyser heats water flowing at the rate of 3.0 litres per minute from 27 °C to 77 °C. If the geyser operates on a gas burner, what is the rate of consumption of the fuel if its heat of combustion is \(4.0 × 10^4\ J/g\) ?
When heat is supplied to a substance, its temperature increases. The amount of heat required to raise the temperature of a given mass of a substance is given by the formula:
\[ Q = mc\Delta T \]
Where:
For water, the specific heat capacity is:
\[ c = 4.2 \times 10^3 \; J\,kg^{-1}°C^{-1} \]
In this problem, the geyser supplies heat to continuously flowing water. The fuel in the gas burner releases heat during combustion. By equating the heat needed by water and the heat released by fuel, we can calculate the fuel consumption rate.
Water enters the geyser at a lower temperature and exits at a higher temperature due to heat supplied by burning fuel.
The density of water is approximately:
\[ 1 \text{ litre of water} \approx 1 \text{ kg} \]
Therefore,
\[ 3 \text{ litres/min} = 3 \text{ kg/min} \]
So, the mass of water heated per minute is:
m = 3 kg/min
\[ \Delta T = T_f - T_i \]
\[ \Delta T = 77 - 27 \]
\[ \Delta T = 50^\circ C \]
Using the heat formula:
\[ Q = mc\Delta T \]
Substitute the values:
\[ Q = 3 \times 4.2 \times 10^3 \times 50 \]
\[ Q = 6.3 \times 10^5 \; J/min \]
So, the geyser must supply 6.3 × 10⁵ Joules of heat every minute.
Heat released by fuel:
\[ 4.0 \times 10^4 \; J/g \]
Fuel required per minute:
\[ \text{Fuel rate} = \frac{\text{Heat required per minute}}{\text{Heat released per gram}} \]
\[ = \frac{6.3 \times 10^5}{4.0 \times 10^4} \]
\[ = 15.75 \; g/min \]
The rate of fuel consumption of the geyser is:
15.75 g per minute
Q2. What amount of heat must be supplied to \(2.0 × 10^{–2}\) kg of nitrogen (at room temperature) to raise its temperature by 45 °C at constant pressure ? (Molecular mass of \(N_2 = 28\); \(R = 8.3\ J\ mol^{-1}\ K^{-1}\).)
When heat is supplied to a gas, its temperature increases. The amount of heat required depends on the number of moles of the gas, its molar heat capacity, and the temperature change.
For a gas heated at constant pressure, the heat supplied is given by:
\[ Q = nC_p\Delta T \]
For a diatomic gas such as nitrogen (\(N_2\)), the molar heat capacity at constant pressure is:
\[ C_p = \frac{7}{2}R \]
where \(R\) is the universal gas constant.
At constant pressure, heat supplied increases the temperature of the gas and allows expansion.
\[ 2.0 × 10^{-2} \text{ kg} = 0.02 \text{ kg} \]
\[ 0.02 \text{ kg} = 20 \text{ g} \]
Number of moles is given by:
\[ n = \frac{\text{mass}}{\text{molar mass}} \]
\[ n = \frac{20}{28} \]
\[ n = 0.714\ \text{mol (approximately)} \]
Since nitrogen is a diatomic gas:
\[ C_p = \frac{7}{2}R \]
\[ C_p = \frac{7}{2} × 8.3 \]
Using the formula:
\[ Q = nC_p\Delta T \]
Substitute the values:
\[ Q = \frac{20}{28} × \frac{7}{2} × 8.3 × 45 \]
Simplifying:
\[ Q = \frac{5}{7} × \frac{7}{2} × 8.3 × 45 \]
\[ Q = 933.75\ J \]
The heat required to raise the temperature of nitrogen gas is:
\(Q \approx 9.34 × 10^2\ J\)
≈ 934 Joules
Q3. Explain why
(a) Two bodies at different temperatures T1 and T2 if brought in thermal contact do
not necessarily settle to the mean temperature (T1 + T2)/2.
(b) The coolant in a chemical or a nuclear plant should have high specific heat.
(c) Air pressure in a car tyre increases during driving.
(d) The climate of a harbour town is more temperate than that of a town in a desert
at the same latitude.
When bodies at different temperatures interact, heat flows from the hotter body to the colder body until thermal equilibrium is reached. The amount of heat exchanged depends on the relation:
\[ Q = mc\Delta T \]
Therefore, the final temperature after heat exchange depends not only on the initial temperatures but also on the heat capacities (mc) of the bodies.
When two bodies at temperatures \(T_1\) and \(T_2\) are brought in contact, heat flows from the hotter body to the colder body until thermal equilibrium is reached.
However, the final temperature depends on the heat capacity of each body:
Heat capacity = \(mc\)
If the bodies have different masses or different specific heat capacities, they exchange different amounts of heat for the same temperature change.
Thus, the equilibrium temperature lies closer to the temperature of the body with the larger heat capacity.
Therefore, the final temperature is not necessarily the simple average \((T_1 + T_2)/2\).
In chemical and nuclear plants, large amounts of heat are produced during reactions. A coolant is used to absorb this excess heat and prevent overheating.
A substance with high specific heat capacity can absorb a large amount of heat without undergoing a large temperature rise.
Therefore, coolants with high specific heat can:
Water is often used as a coolant because it has a very high specific heat capacity.
While driving, the tyre continuously deforms where it touches the road. This causes:
These processes increase the internal energy of the air inside the tyre, raising its temperature.
According to the gas law:
\[ P \propto T \quad (\text{for nearly constant volume}) \]
Since the tyre volume changes very little, an increase in temperature causes the air pressure to increase.
Harbour towns are located near large water bodies such as seas or oceans. Water has a high specific heat capacity.
Because of this:
This slow heating and cooling moderates the temperature of nearby land areas.
In contrast, desert regions have sand with low specific heat capacity. Sand heats and cools rapidly, causing large temperature variations.
Therefore, harbour towns experience a more moderate (temperate) climate compared to desert towns at the same latitude.
Q4. A cylinder with a movable piston contains 3 moles of hydrogen at standard temperature and pressure. The walls of the cylinder are made of a heat insulator, and the piston is insulated by having a pile of sand on it. By what factor does the pressure of the gas increase if the gas is compressed to half its original volume ?
When a gas is compressed or expanded without any heat exchange with the surroundings, the process is called an adiabatic process.
In an adiabatic process, pressure and volume are related by:
\[ PV^{\gamma} = \text{constant} \]
or
\[ P_1 V_1^{\gamma} = P_2 V_2^{\gamma} \]
Where:
For a diatomic gas such as hydrogen:
\[ \gamma = \frac{7}{5} = 1.4 \]
During adiabatic compression, volume decreases while pressure and temperature increase.
\[ P_1 V_1^{\gamma} = P_2 V_2^{\gamma} \]
Rearranging to find the pressure ratio:
\[ \frac{P_2}{P_1} = \left(\frac{V_1}{V_2}\right)^{\gamma} \]
The gas is compressed to half of its original volume:
\[ V_2 = \frac{V_1}{2} \]
Therefore,
\[ \frac{V_1}{V_2} = 2 \]
Substitute the value of \(\gamma = 1.4\):
\[ \frac{P_2}{P_1} = 2^{1.4} \]
\[ \frac{P_2}{P_1} \approx 2.64 \]
When the gas is compressed to half of its original volume under adiabatic conditions, the pressure increases by a factor of:
≈ 2.64
Q5. In changing the state of a gas adiabatically from an equilibrium state A to another equilibrium state B, an amount of work equal to 22.3 J is done on the system. If the gas is taken from state A to B via a process in which the net heat absorbed by the system is 9.35 cal, how much is the net work done by the system in the latter case ? (Take 1 cal = 4.19 J)
The relationship between heat, work, and internal energy of a system is given by the First Law of Thermodynamics:
\[ \Delta U = Q - W \]
Important points:
Heat supplied to a system is used to change its internal energy and perform work.
During the first process (adiabatic):
Since work done on the system is negative work by the system:
\[ W = -22.3\; J \]
Using the first law:
\[ \Delta U = Q - W \]
\[ \Delta U = 0 - (-22.3) \]
\[ \Delta U = 22.3\; J \]
Thus, the change in internal energy between states A and B is:
ΔU = 22.3 J
Heat absorbed in the second process:
\[ Q_2 = 9.35\; cal \]
Using the conversion:
\[ 1\; cal = 4.19\; J \]
\[ Q_2 = 9.35 \times 4.19 \]
\[ Q_2 \approx 39.2\; J \]
Using:
\[ \Delta U = Q_2 - W \]
Substitute the known values:
\[ 22.3 = 39.2 - W \]
Solving for \(W\):
\[ W = 39.2 - 22.3 \]
\[ W = 16.9\; J \]
The net work done by the system in the second process is:
16.9 J
Q6. Two cylinders A and B of equal capacity are connected to each other via a stopcock.
A contains a gas at standard temperature and pressure. B is completely evacuated.
The entire system is thermally insulated. The stopcock is suddenly opened. Answer
the following :
(a) What is the final pressure of the gas in A and B ?
(b) What is the change in internal energy of the gas ?
(c) What is the change in the temperature of the gas ?
(d) Do the intermediate states of the system lie on its P–V–T surface ?
When a gas expands suddenly into a vacuum without external resistance, the process is called free expansion.
Important properties of free expansion:
\[ \Delta U = Q - W \]
Thus in free expansion:
\[ \Delta U = 0 \]
For an ideal gas, internal energy depends only on temperature. Therefore, if internal energy does not change, the temperature also remains constant.
Gas expands freely from cylinder A into the evacuated cylinder B when the stopcock is opened.
Initially:
After opening the stopcock, the gas spreads into both cylinders.
Final volume of gas:
\[ V_f = V + V = 2V \]
Since temperature remains constant, using the ideal gas relation:
\[ P_1 V = P_f (2V) \]
\[ P_f = \frac{P_1}{2} \]
Therefore, the final pressure in both cylinders becomes:
Final Pressure = \(P_1 / 2\)
Using the First Law of Thermodynamics:
\[ \Delta U = Q - W \]
Since the system is insulated:
\[ Q = 0 \]
Expansion occurs into vacuum, so:
\[ W = 0 \]
Therefore:
\[ \Delta U = 0 \]
Change in Internal Energy = 0
For an ideal gas:
Internal Energy \(U \propto T\)
Since internal energy does not change:
\[ \Delta T = 0 \]
Temperature remains unchanged.
The P–V–T surface represents only equilibrium states of a gas.
During free expansion:
Therefore, these intermediate states cannot be represented by a single point on the P–V–T surface.
Hence, intermediate states do not lie on the P–V–T surface.
Q7. An electric heater supplies heat to a system at a rate of 100 W. If the system performs work at a rate of 75 joules per second, at what rate is the internal energy increasing?
The First Law of Thermodynamics states that the heat supplied to a system is used to increase its internal energy and to perform work.
\[ \Delta U = Q - W \]
For processes where heat and work occur continuously, we use the rate form of the first law:
\[ \frac{dU}{dt} = \dot{Q} - \dot{W} \]
Note: 1 watt (W) = 1 joule per second (J/s).
Heat supplied to a system is partly converted into work and partly increases internal energy.
Since \(1 W = 1 J/s\):
\[ \dot{Q} = 100\ J/s \]
\[ \dot{W} = 75\ J/s \]
\[ \frac{dU}{dt} = \dot{Q} - \dot{W} \]
\[ \frac{dU}{dt} = 100 - 75 \]
\[ \frac{dU}{dt} = 25\ J/s \]
Since \(1\ J/s = 1\ W\):
\[ \frac{dU}{dt} = 25\ W \]
The internal energy of the system is increasing at a rate of:
25 W (or 25 J/s)
Q8. A thermodynamic system is taken from an original state to an intermediate state by
the linear process shown in Fig. (11.11). Its volume is then reduced to the original value
from E to F by an isobaric process.
Calculate the total work done by the gas from D → E → F.
In thermodynamics, the work done by a gas during expansion or compression can be determined from a Pressure–Volume (P–V) diagram.
\[ W = \int P\, dV \]
Graphically, this means:
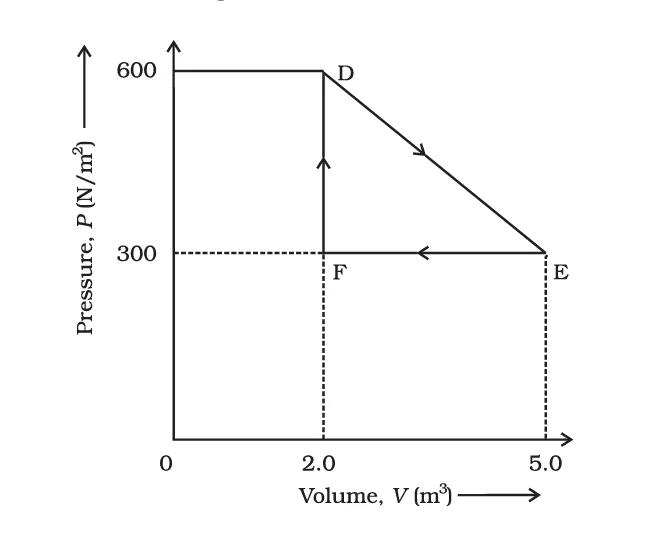
The process from D to E is a linear process on the P–V diagram. Therefore, the work done equals the area under the straight line DE.
This area forms a trapezium.
Given:
\[ W_{DE} = \frac{1}{2}(P_D + P_E)(V_E - V_D) \]
\[ W_{DE} = \frac{1}{2}(600 + 300)(5 - 2) \]
\[ W_{DE} = \frac{1}{2} \times 900 \times 3 \]
\[ W_{DE} = 1350\ J \]
Since the volume increases, the gas expands, so the work done is positive.
The process from E to F is an isobaric process (constant pressure).
Pressure:
\[ P = 300\ N\,m^{-2} \]
Volume decreases from:
Work done in an isobaric process:
\[ W = P(V_F - V_E) \]
\[ W_{EF} = 300(2 - 5) \]
\[ W_{EF} = -900\ J \]
The negative sign indicates compression (work done on the gas).
Total work done during the process D → E → F:
\[ W_{total} = W_{DE} + W_{EF} \]
\[ W_{total} = 1350 - 900 \]
\[ W_{total} = 450\ J \]
Total work done by the gas = 450 J
Get in Touch
Questions, feedback, or suggestions?
We'd love to hear from you.