From the Zeroth Law to Carnot's engine — rigorous notes, derivations, PV diagrams, and exam-ready key points for CBSE Boards, JEE & NEET.
Thermal equilibrium is the state of a thermodynamic system in which there is no net flow of heat either within the system or between the system and its surroundings. When this condition is reached, the temperature throughout the system becomes uniform and constant with time.
In simple terms, when two or more bodies are placed in thermal contact and their macroscopic thermal properties such as temperature stop changing, the bodies are said to be in thermal equilibrium.
Temperature determines the direction of heat flow. According to thermodynamic principles, heat always flows spontaneously from a region of higher temperature to a region of lower temperature.
Thermal equilibrium is achieved when the temperature difference between interacting systems becomes zero:
\[ \Delta T = 0 \]At this stage, the driving force responsible for heat transfer disappears, and therefore no further heat exchange occurs.
The concept of thermal equilibrium forms the foundation of the Zeroth Law of Thermodynamics, which states:
"If two systems are separately in thermal equilibrium with a third system, then they are in thermal equilibrium with each other."
This law provides the basis for the measurement of temperature using thermometers.
The Zeroth Law of Thermodynamics establishes the fundamental concept of temperature and thermal equilibrium between physical systems.
Law: If two thermodynamic systems are each in thermal equilibrium with a third system, then the two systems are also in thermal equilibrium with each other.
Although this principle appears simple, it is extremely important because it provides the logical basis for measuring temperature. Interestingly, this law was formulated after the First and Second Laws of Thermodynamics, which is why it was named the "Zeroth Law".
The Zeroth Law tells us that thermal equilibrium is a transitive property. In other words, if two systems independently show no heat exchange with a third system, they must share the same thermal condition.
This implies the existence of a unique physical property known as temperature.
Consider three systems: A, B, and C.
If system A is in thermal equilibrium with system C:
\[ T_A = T_C \]And system B is also in thermal equilibrium with system C:
\[ T_B = T_C \]Then it directly follows that:
\[ T_A = T_B \]The common quantity T represents the thermodynamic property called temperature.
Thus, temperature is defined not by heat itself, but by the condition of thermal equilibrium.
The Zeroth Law explains how a thermometer measures temperature.
Thus, the thermometer acts as the third system in the Zeroth Law.
Without the Zeroth Law, the concept of temperature would remain undefined and thermodynamics would lack a consistent experimental framework.
Heat is the form of energy that is transferred from one physical system to another solely because of a difference in temperature.
Unlike internal energy, heat is not stored within a body. It exists only during the process of energy transfer between systems that are at different temperatures.
The amount of heat transferred during a temperature change is often expressed as:
\[ Q = mc\Delta T \]where:
When a hot cup of tea is left in a cool room, heat flows from the tea to the surrounding air until both reach the same temperature. At that point, thermal equilibrium is achieved.
Internal energy is the total microscopic energy possessed by a thermodynamic system due to the random motion and interactions of its constituent particles.
It represents the energy stored within the system at the molecular level.
Internal energy consists mainly of two contributions:
At the molecular level, particles in matter are constantly moving and interacting. The total energy of all these microscopic motions and interactions collectively forms the internal energy (U) of the system.
For an ideal gas, internal energy depends mainly on temperature.
\[ U \propto T \]In thermodynamics, work refers to the transfer of energy that occurs when an external force causes a macroscopic displacement of the boundary of a system.
Unlike heat, which arises due to temperature difference, work results from mechanical interaction between the system and its surroundings.
Consider a gas enclosed in a cylinder fitted with a movable piston.
When a gas expands against an external pressure, the infinitesimal work done is given by:
\[ dW = P\,dV \]For a finite change in volume:
\[ W = \int P\,dV \]where:
The First Law of Thermodynamics is a direct consequence of the law of conservation of energy.
The change in internal energy of a system is equal to the heat supplied to the system minus the work done by the system on its surroundings.
Mathematically, the law is expressed as:
\[ \Delta U = Q - W \]where
The First Law explains that energy supplied to a system is not lost. It is simply redistributed into two forms:
If heat \(Q\) is supplied to a system:
\[ Q = \Delta U + W \]This means the supplied heat is partly used to increase the internal energy of the system and partly used to perform mechanical work.
Let
According to conservation of energy:
\[ \Delta Q = \Delta U + \Delta W \]This equation shows that the energy supplied to a system is distributed between increasing internal energy and performing work on the surroundings.
These limitations are addressed by the Second Law of Thermodynamics.
The specific heat capacity of a substance is the amount of heat required to raise the temperature of unit mass of the substance by one degree without changing its physical state.
It represents how much thermal energy a material can store. Substances with high specific heat capacity require a large amount of heat to change their temperature.
If an amount of heat \(Q\) is supplied to a body of mass \(m\), causing its temperature to change by \(\Delta T\), the specific heat capacity is defined as:
\[ s = \frac{Q}{m\Delta T} \]where
The SI unit of specific heat capacity is:
\[ \text{J kg}^{-1}\text{K}^{-1} \]Suppose an amount of heat \(\Delta Q\) is supplied to a substance, raising its temperature from \(T\) to \(T+\Delta T\).
The heat capacity \(S\) of the substance is defined as:
\[ S=\frac{\Delta Q}{\Delta T} \]Heat capacity depends on the total amount of substance present. Therefore, it is proportional to the mass \(m\).
To obtain a property independent of the quantity of material, we divide heat capacity by mass:
\[ s=\frac{S}{m} \] \[ s=\frac{1}{m}\frac{\Delta Q}{\Delta T} \]This quantity \(s\) is called the specific heat capacity.
If the amount of substance is expressed in terms of the number of moles \(\mu\), the heat capacity per mole is defined as:
\[ C=\frac{S}{\mu} \] \[ C=\frac{1}{\mu}\frac{\Delta Q}{\Delta T} \]\(C\) is called the molar specific heat capacity.
Its SI unit is:
\[ \text{J mol}^{-1}\text{K}^{-1} \]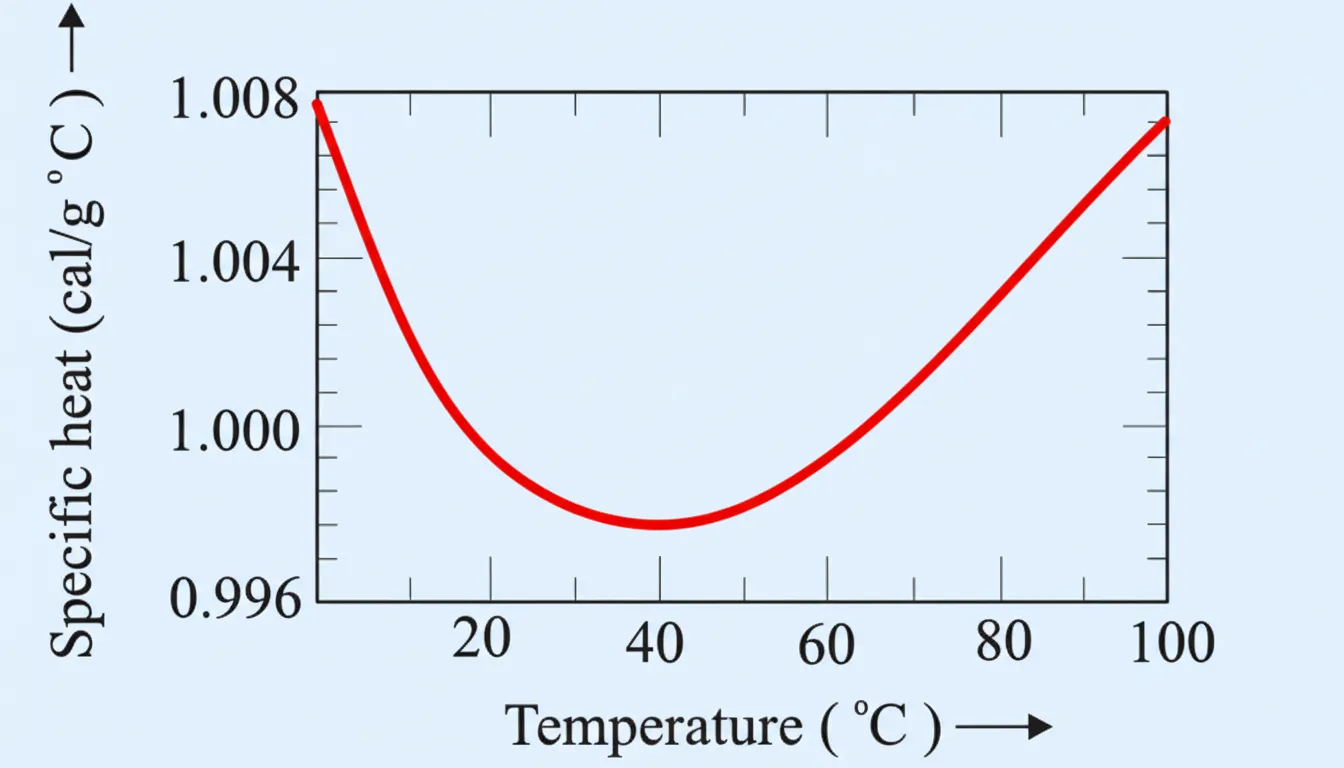
The traditional unit used to measure heat was the calorie. Originally, one calorie was defined as the amount of heat required to raise the temperature of 1 gram of water by 1 °C.
Water was chosen as the reference substance because it is easily available and exhibits highly reproducible thermal behaviour. This definition therefore provided a convenient experimental standard for early calorimetric measurements.
More precise experiments later showed that the specific heat capacity of water is not perfectly constant. Instead, it changes slightly with temperature.
Careful calorimetric studies indicate that the heat required to increase the temperature of water by 1 °C depends on the initial temperature of the water. This variation occurs over the temperature range from 0 °C to 100 °C, as illustrated in Fig. 11.5.
Because of the temperature dependence of specific heat capacity, the original definition of calorie became ambiguous. To remove this uncertainty, a precise temperature interval was adopted.
Modern definition: One calorie is defined as the amount of heat required to raise the temperature of 1 g of water from 14.5 °C to 15.5 °C under standard atmospheric conditions.
In modern physics, heat is treated as a form of energy transfer. Therefore, it is convenient to express heat in the SI unit of energy: the joule (J).
The specific heat capacity of water in SI units is
\[ \begin{aligned} c_{\text{water}} &= 4186 \ \text{J kg}^{-1}\text{K}^{-1} \\\\ &= 4.186 \ \text{J g}^{-1}\text{K}^{-1} \end{aligned} \]This high value explains why water can absorb or release large amounts of heat with only a small change in temperature.
Historically, scientists compared heat with mechanical work through a quantity called the mechanical equivalent of heat.
It represented the amount of mechanical work required to produce one calorie of heat.
In modern thermodynamics this concept simply acts as a conversion factor between two units of energy (calorie and joule). Since the SI system uses joules for all forms of energy, the term has largely lost its independent significance.
A thermodynamic state variable is a physical quantity whose value depends only on the current state of a system and not on the process or path by which the system reached that state.
These variables provide a complete macroscopic description of the system when it is in thermodynamic equilibrium.
Common examples of thermodynamic state variables include:
A key property of state variables is that the change in their value between two states depends only on the initial and final states, not on the thermodynamic path connecting them.
Thermodynamic state variables are broadly classified into two categories:
This distinction plays a crucial role in thermodynamic analysis and in developing mathematical relations such as the equation of state.
A system is said to be in thermodynamic equilibrium when it simultaneously satisfies three equilibrium conditions:
Only under thermodynamic equilibrium can the state of a system be uniquely described using state variables.
An equation of state is a mathematical relation that connects the thermodynamic state variables of a system.
For an ideal gas, experimental observations show that:
\[ \frac{PV}{T} = \text{constant} \]For one mole of an ideal gas, this constant is denoted by the universal gas constant \(R\), giving
\[ PV = RT \]For \(\mu\) moles of gas, the equation becomes
\[ \boxed{PV = \mu RT} \]Thermodynamic state variables provide a bridge between the microscopic behaviour of molecules and the macroscopic properties of matter.
The equation of state reduces the number of independent variables needed to describe a system and allows physicists to analyze thermodynamic processes mathematically.
Understanding state variables is essential for studying:
Thus, the equation of state forms one of the fundamental mathematical foundations of thermodynamics.
A quasi-static process is a thermodynamic process that occurs so slowly that the system remains in thermodynamic equilibrium at every intermediate stage of the process.
Although the system is continuously changing, it moves through a continuous sequence of equilibrium states. Because of this, all thermodynamic state variables such as pressure (P), volume (V), and temperature (T) are well defined throughout the process.
The word quasi-static literally means “almost static”.
In a perfectly quasi-static process, the system evolves infinitely slowly, ensuring that the difference between the system and the surroundings remains extremely small.
In practice, a perfectly quasi-static process is impossible because it would require infinite time to complete. However, many real processes can be approximated as quasi-static when they occur sufficiently slowly.
Consider a gas enclosed in a cylinder fitted with a movable piston. If the external pressure is increased in extremely small steps, the gas compresses very slowly.
A quasi-static process maintains equilibrium conditions because:
An isothermal process is a thermodynamic process in which the temperature of the system remains constant throughout the process.
Mathematically,
\[ \Delta T = 0 \]In such a process, the system continuously exchanges heat with its surroundings so that the temperature does not change despite expansion or compression.
For a process to remain isothermal, two important conditions must be satisfied:
Rapid processes generally do not allow sufficient time for heat transfer and therefore cannot maintain constant temperature.
For an ideal gas, the equation of state is
\[ PV = \mu RT \]Since temperature \(T\) remains constant during an isothermal process,
\[ PV = \text{constant} \]This relation is known as Boyle's Law. It implies that pressure is inversely proportional to volume.
Consider \(\mu\) moles of an ideal gas expanding isothermally from volume \(V_1\) to \(V_2\) at temperature \(T\).
At any intermediate stage,
\[ P = \frac{\mu RT}{V} \]The infinitesimal work done by the gas during expansion \(dV\) is
\[ dW = P\,dV = \frac{\mu RT}{V} dV \]Integrating between \(V_1\) and \(V_2\),
\[ \begin{aligned} W &= \int_{V_1}^{V_2} \frac{\mu RT}{V} dV \\\\ &= \mu RT \ln \left(\frac{V_2}{V_1}\right) \end{aligned} \]This expression represents the work done by an ideal gas during a quasi-static isothermal expansion.
For an ideal gas, the internal energy depends only on temperature. Since temperature remains constant,
\[ \Delta U = 0 \]Applying the First Law of Thermodynamics
\[ \Delta Q = \Delta U + W \]Substituting \(\Delta U = 0\),
\[ \Delta Q = W \]Thus, the heat absorbed by the gas is entirely converted into work done by the gas.
Continuous heat exchange with the surroundings is therefore a defining feature of an isothermal process.
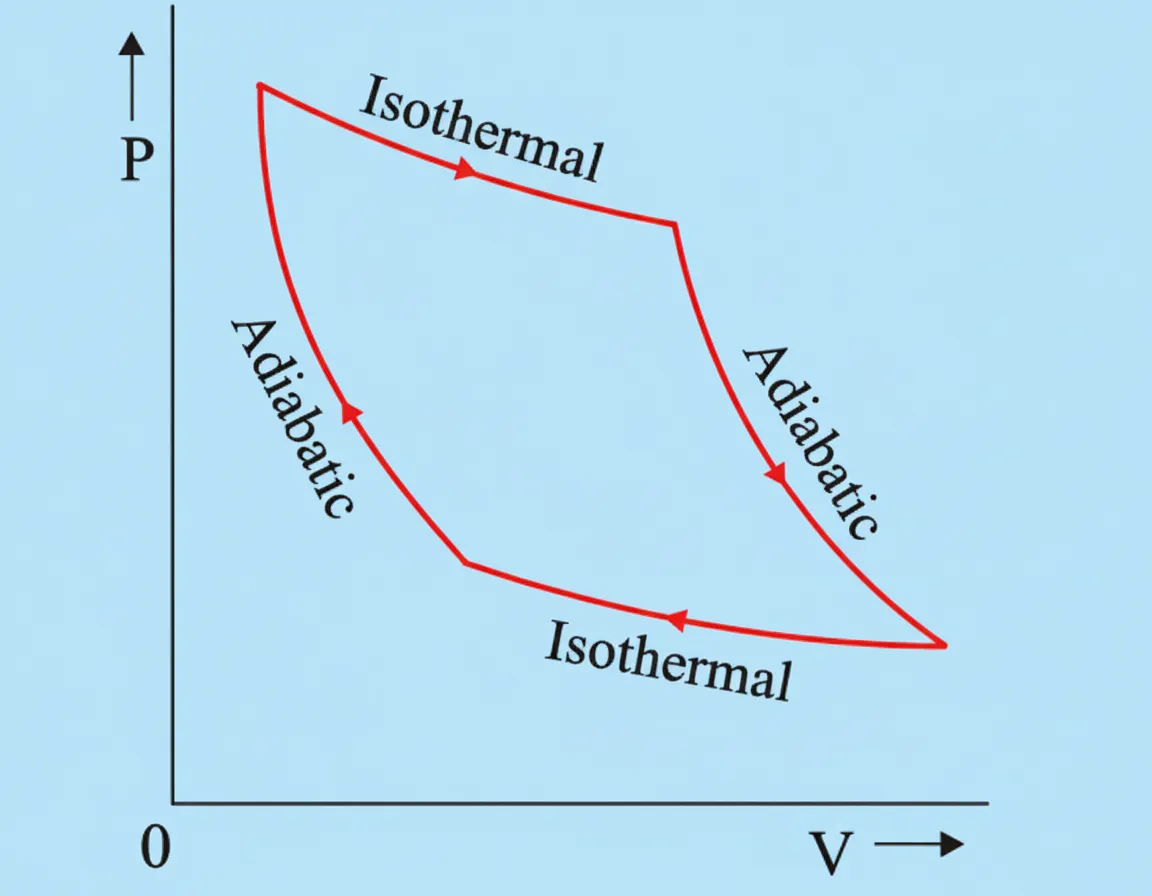
An adiabatic process is a thermodynamic process in which no heat is exchanged between the system and its surroundings.
Mathematically,
\[ \Delta Q = 0 \]In such a process, any change in the internal energy of the system occurs solely due to work done by or on the system.
An adiabatic process can occur under two conditions:
Rapid compression of air in a bicycle pump is a common real-life example of an approximately adiabatic process.
For an ideal gas undergoing an adiabatic process, the pressure and volume are related by
\[ PV^{\gamma} = \text{constant} \]where
\[ \gamma = \frac{C_p}{C_v} \]is called the ratio of specific heats.
If a gas changes from state \((P_1, V_1)\) to \((P_2, V_2)\), then
\[ P_1 V_1^{\gamma} = P_2 V_2^{\gamma} \]Work done during a quasi-static adiabatic change is
\[ W = \int_{V_1}^{V_2} P \, dV \]Using the relation \(PV^{\gamma} = \text{constant}\), the work done becomes
\[ W = \frac{P_1 V_1 - P_2 V_2}{\gamma - 1} \]Using the ideal gas equation \(PV=\mu RT\), this can also be written as
\[ W = \frac{\mu R (T_1 - T_2)}{\gamma - 1} \]This expression gives the work done by an ideal gas during an adiabatic expansion or compression.
On a P–V diagram, an adiabatic curve is steeper than the corresponding isothermal curve passing through the same initial point.
This occurs because during adiabatic expansion, the temperature of the gas decreases, causing the pressure to fall more rapidly with increasing volume.
An isochoric process is a thermodynamic process in which the volume of the system remains constant throughout the process.
Mathematically,
\[ \Delta V = 0 \]Such a process generally occurs when a gas is confined in a rigid container that does not allow expansion or compression.
Since the volume of the system does not change, the boundary of the system does not move. Therefore, the system cannot perform mechanical work on the surroundings.
However, the temperature and pressure of the system may change due to heat transfer.
Work done during a thermodynamic process is given by
\[ W = \int P\, dV \]Since the volume remains constant during an isochoric process,
\[ dV = 0 \]Therefore,
\[ W = 0 \]This shows that no mechanical work is done during an isochoric process.
The first law of thermodynamics is expressed as
\[ \Delta Q = \Delta U + W \]For an isochoric process, since \(W = 0\),
\[ \Delta Q = \Delta U \]This result implies that all the heat supplied to the system is used entirely to change its internal energy.
For an ideal gas, the equation of state is
\[ PV = \mu RT \]If the volume remains constant, the relation becomes
\[ \frac{P}{T} = \text{constant} \]Thus, in an isochoric process, the pressure of the gas is directly proportional to its temperature.
The First Law of Thermodynamics establishes the principle of energy conservation. However, it does not specify the direction in which thermodynamic processes occur.
For example, the first law does not explain:
These limitations are addressed by the Second Law of Thermodynamics, which introduces the concepts of irreversibility and directionality in natural processes.
The second law explains why certain processes occur naturally while their reverse cannot occur without external intervention.
For instance:
Thus, the second law determines the natural direction of thermodynamic processes.
One of the most important formulations of the second law of thermodynamics is the Kelvin–Planck statement.
Kelvin–Planck Statement:
It is impossible to construct a device that operates in a cyclic process
and produces no effect other than the absorption of heat from a single
thermal reservoir and the complete conversion of that heat into work.
In simple terms, no heat engine can convert all the heat absorbed from a single reservoir entirely into work. Some heat must always be rejected to a lower-temperature reservoir.
A practical heat engine operates between two thermal reservoirs:
Only a portion of the heat absorbed from the hot reservoir can be converted into useful work. The remaining heat must be rejected to the cold reservoir.
Another important formulation of the Second Law of Thermodynamics is given by the Clausius statement.
Clausius Statement:
It is impossible to construct a device that operates in a cyclic process
and has no effect other than the transfer of heat from a colder body to
a hotter body.
In simpler terms, heat cannot spontaneously flow from a colder body to a hotter body without the assistance of external work.
Natural heat flow always occurs from a region of higher temperature to lower temperature. This spontaneous direction of heat transfer is a direct consequence of the second law of thermodynamics.
If heat must be transferred from a cold body to a hot body, external work must be supplied. This is exactly how refrigeration and air-conditioning systems operate.
A refrigerator transfers heat from the cold interior compartment to the warmer surroundings. According to the Clausius statement, this transfer cannot occur naturally.
Therefore, a refrigerator requires external work supplied by an electric motor to pump heat from the cold region to the hot environment.
In thermodynamics, not all processes occur with the same degree of symmetry or reversibility. Some processes can be reversed so that both the system and its surroundings return exactly to their original states, while others cannot be undone without leaving permanent changes.
This leads to the classification of thermodynamic processes into reversible and irreversible processes. Understanding this distinction is fundamental for analysing natural processes and for applying the second law of thermodynamics.
A reversible process is a thermodynamic process that can be reversed by an infinitesimal change in external conditions so that both the system and the surroundings are restored exactly to their initial states.
During a reversible process, the system passes through a continuous sequence of equilibrium states. Because this requires infinitely slow changes and the complete absence of dissipative effects, reversible processes are idealised theoretical models rather than real physical processes.
For a thermodynamic process to be reversible, the following conditions must be satisfied:
If any of these conditions is violated, the process becomes irreversible.
For a reversible expansion of a gas, the external pressure differs from the internal pressure of the gas by an infinitesimally small amount. Therefore, the pressure is well defined at every stage.
The work done during the process is given by
\[ W=\int P\,dV \]Because the system remains in equilibrium throughout the process, this expression gives the maximum possible work obtainable between two given thermodynamic states.
An irreversible process is a thermodynamic process that cannot be reversed without producing permanent changes in the system or its surroundings.
Most real processes occurring in nature are irreversible because they involve finite driving forces such as temperature differences, pressure differences, friction, or turbulence.
Examples include free expansion of gases, heat transfer through a finite temperature difference, and mechanical motion with friction.
The following table summarises the major differences between reversible and irreversible thermodynamic processes:
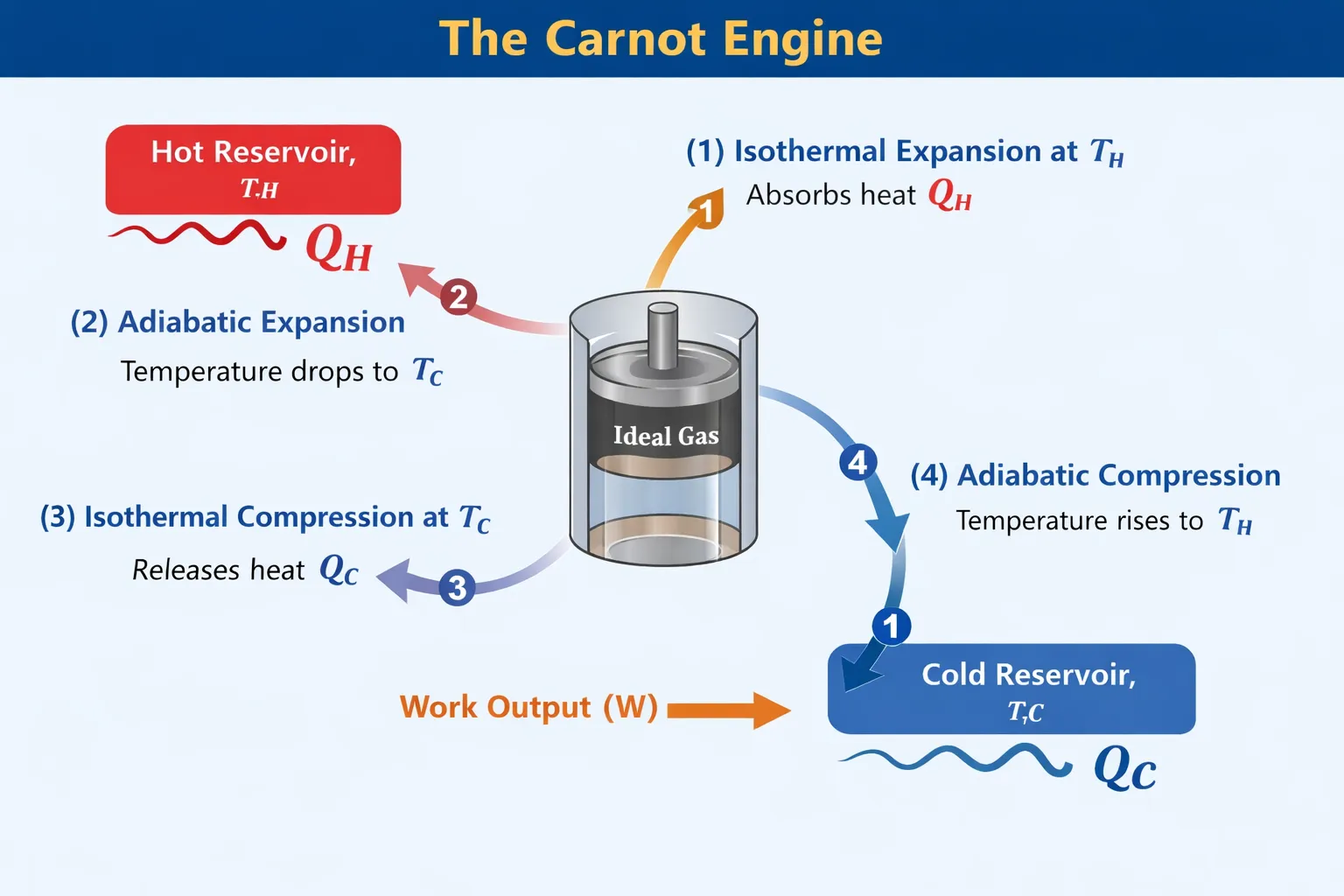
A Carnot engine is an ideal heat engine that operates in a completely reversible cycle between two thermal reservoirs at fixed temperatures and converts heat into work with the maximum possible efficiency.
It operates between two reservoirs:
where
\[ T_H > T_C \]The Carnot engine represents an idealized model used to determine the upper limit of efficiency for all heat engines.
The Carnot engine consists of an ideal gas enclosed in a perfectly insulated cylinder fitted with a frictionless piston.
The cylinder can be placed alternately in contact with:
All processes occur quasi-statically to maintain thermodynamic equilibrium and ensure complete reversibility.
The Carnot engine operates through a cycle consisting of four reversible thermodynamic processes.
The efficiency of any heat engine is defined as
\[ \eta = \frac{W}{Q_H} \]where
For a cyclic process,
\[ W = Q_H - Q_C \]Therefore,
\[ \eta = 1 - \frac{Q_C}{Q_H} \]For a reversible Carnot cycle,
\[ \frac{Q_C}{Q_H} = \frac{T_C}{T_H} \]Hence the efficiency becomes
\[ \boxed{\eta = 1 - \frac{T_C}{T_H}} \]This important result shows that the efficiency of a Carnot engine depends only on the temperatures of the reservoirs and not on the nature of the working substance.
Get in Touch
Questions, feedback, or suggestions?
We'd love to hear from you.