Structure of Atom is where modern chemistry begins. It explains why elements behave the way they do — their bonding, reactivity, and every property you'll study hereafter traces back to this chapter.
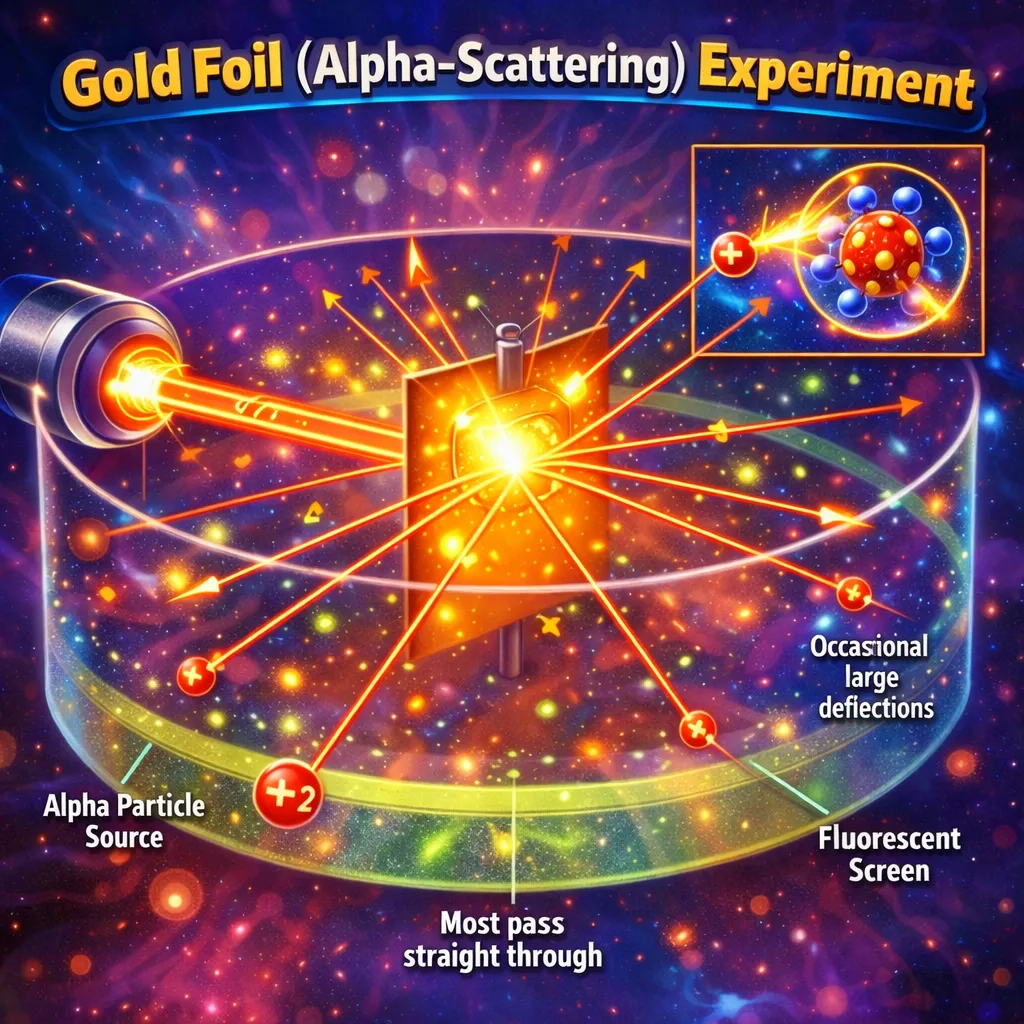
What is matter ultimately made of? Philosophers asked this question for millennia, but it took centuries of experiments — cathode rays, gold foil, spectral lines — to finally answer it. NCERT Class XI Chemistry Chapter 2 takes you through that journey: from the discovery of electrons and protons to quantum mechanical orbitals, building up a picture of the atom that is both beautifully logical and deeply counterintuitive.
What This Chapter Covers
You begin with the discovery of subatomic particles — the electron (Thomson), proton (Rutherford), and neutron (Chadwick) — and learn how each was identified through landmark experiments.
The chapter then walks through the evolution of atomic models: Thomson's plum pudding, Rutherford's nuclear model, Bohr's quantised orbits, and finally the quantum mechanical model. Each model corrected the flaws of the last.
A pivotal section covers atomic spectra — why hydrogen emits specific lines of light — and how Bohr's model explained them using quantised energy levels. You'll learn to calculate the energy and wavelength of emitted photons.
The chapter climaxes with quantum mechanics: Heisenberg's uncertainty principle, de Broglie's wave-particle duality, and the concept of orbitals described by quantum numbers. Electronic configurations and the Aufbau, Pauli, and Hund rules complete the picture.
Key Takeaways
How to Study This Chapter
Don't jump straight to quantum numbers. Study the experiments in order — cathode rays → gold foil → spectral lines. Each experiment revealed a flaw in the prevailing model and forced a new one. Understanding why each model was proposed makes the quantum leap feel logical, not arbitrary.
Energy levels, wavelengths of spectral lines, and the Rydberg formula are high-frequency numerical topics. Practice calculating energy transitions for hydrogen and hydrogen-like ions. Know the spectral series — Lyman (UV), Balmer (visible), Paschen (IR) — by heart.
Learn what each quantum number physically means: n (energy/shell), l (shape/subshell), ml (orientation), ms (spin). Draw the shapes of s, p, and d orbitals. The rules for allowed values (l < n, ml ranges from −l to +l) follow naturally once you understand what you're describing.
Write configurations for the first 30 elements without looking. Pay special attention to exceptions like Cr (3d⁵ 4s¹) and Cu (3d¹⁰ 4s¹) — half-filled and fully-filled subshells are extra stable. These exceptions appear in exams every year.
Most students struggle with quantum numbers because they try to memorise rules without understanding them. Spend one focused session on just the physical meaning of n, l, ml, and ms. Once those click, electronic configuration becomes a logical exercise — not a memory task. That session is worth five hours of rote memorisation.
Ready to go deeper?
Everything you need to master Structure of Atom — from first read to exam day.